Research ► Materials Thermodynamics
Materials Thermodynamics
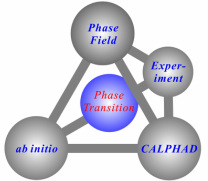
Studying phase behavior in materials needs an integrated method with inputs from both experiments and modeling. In my research projects, I concentrate on the ab initio, CALPHAD and phase field methods for simulation. Meanwhile, the designed key experiments are also employed to validate the simulated results.
1.1. High quality thermodynamic database for industry (Application driven research)
I have made considerable contributions to several different industrial thermodynamic/diffusion databases. Most of them are now commercialized as the Thermo-Calc databases, such as TCAL for Al research, TCNI for Ni superalloys design.
1.2. Development of the CALPHAD approach. Performing thermodynamic modeling done to 0 K !
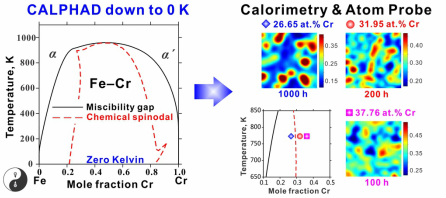
Thermodynamic description on the Fe-Cr system as a case study was published in the CALPHAD journal, and won the CALPHAD Best Paper Award in 2012 as the best manuscript published in the CALPHAD journal in 2011.
1.3. Fundamental model for the CALPHAD approach coupling with the ab initio calculations: a promising way to bridge CALPHAD and ab-initio calculations.
I am involved in developing the next generation lattice stability database for the CALPHAD approach. Please check this paper (link) to see more details. In another word, this work will enable CALPHAD models to predict thermodynamic properties at low temperature approaching to zero Kelvin. Therefore, CALPHAD can be expected to seamlessly integrate with the DFT quantum mechanical calculations. This will bring more opportunities to create reliable thermodynamic database for advanced materials and processing design over a much wider composition range.
1.4. Phase stability of intermetallics
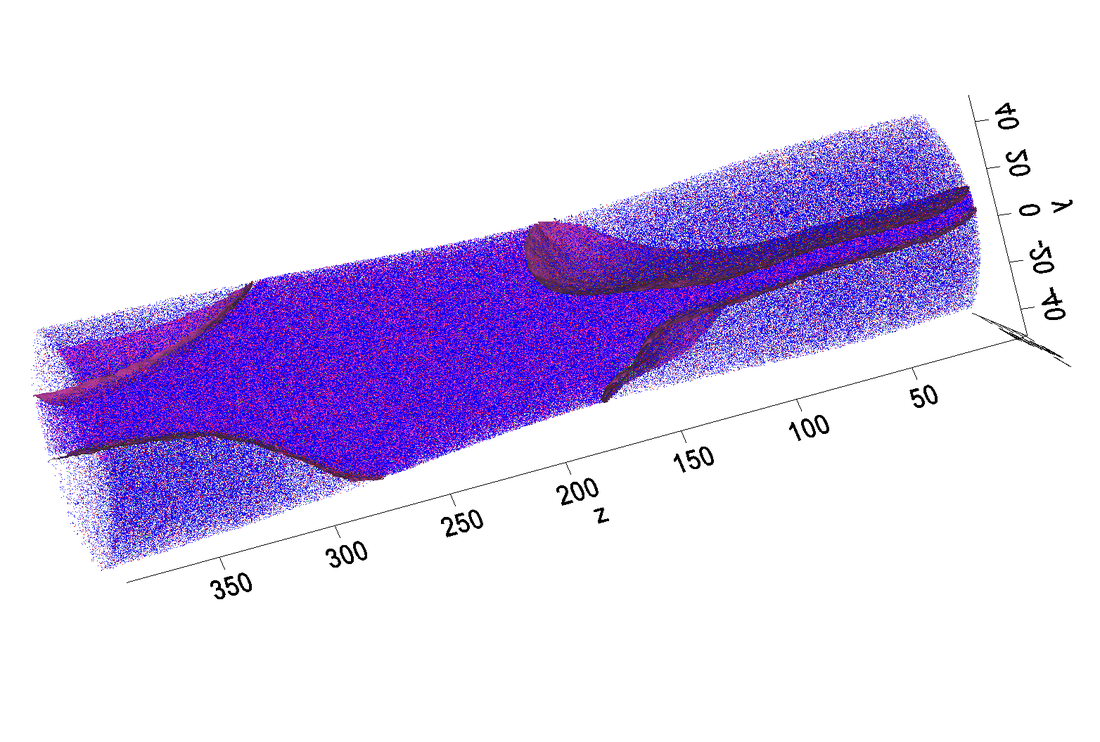
Atom probe tomography is a powerful tool to assist materials design. In my project, LEAP is used for studying nano-precipitation and spinodal decomposition in different alloy systems. Two examples are given here. one is Q phase precipitation in Al alloy, and the other is grain boundary analysis of the newly designed Ni-superalloy. Figures shown below are in nano-scale. nm as unit.
I have made considerable contributions to several different industrial thermodynamic/diffusion databases. Most of them are now commercialized as the Thermo-Calc databases, such as TCAL for Al research, TCNI for Ni superalloys design.
1.2. Development of the CALPHAD approach. Performing thermodynamic modeling done to 0 K !
Thermodynamic description on the Fe-Cr system as a case study was published in the CALPHAD journal, and won the CALPHAD Best Paper Award in 2012 as the best manuscript published in the CALPHAD journal in 2011.
1.3. Fundamental model for the CALPHAD approach coupling with the ab initio calculations: a promising way to bridge CALPHAD and ab-initio calculations.
I am involved in developing the next generation lattice stability database for the CALPHAD approach. Please check this paper (link) to see more details. In another word, this work will enable CALPHAD models to predict thermodynamic properties at low temperature approaching to zero Kelvin. Therefore, CALPHAD can be expected to seamlessly integrate with the DFT quantum mechanical calculations. This will bring more opportunities to create reliable thermodynamic database for advanced materials and processing design over a much wider composition range.
1.4. Phase stability of intermetallics
Atom probe tomography is a powerful tool to assist materials design. In my project, LEAP is used for studying nano-precipitation and spinodal decomposition in different alloy systems. Two examples are given here. one is Q phase precipitation in Al alloy, and the other is grain boundary analysis of the newly designed Ni-superalloy. Figures shown below are in nano-scale. nm as unit.
1.5. Experimental investigation of the phase separation and precipitations of detrimental phases in the Fe-Cr-X systems
|
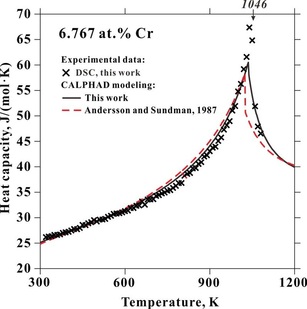
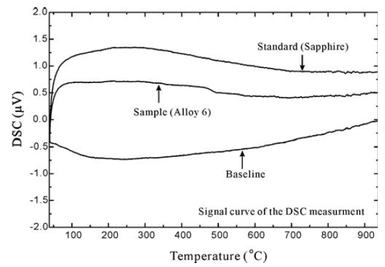
It is fundamental to determine thermodynamic properties of the alloys when studying phase transformations. One of these properties is heat capacity which could be determined from Differential Scanning Calorimetry (DSC). The left column shows one determined heat capacity of the Fe-Cr binary alloy. One shall noticed that a determination of the heat capacity needs patience. Especially for achieving good baseline which will affect the accuracy of the final results quite often. The derivation of the heat capacity could be defined by using the follow equation. Sapphire is one of the good standard used for alloy systems since its Cp is well know already. As we can find from the left bottom figure which shows original signal from the DSC measurement. One shall keep in mind that the baseline running with sample and empty crucible should be kept as close as possible in order to achieve reliable results. Because adding sample in to crucible could easily move the position of the crucible.
More related content are discussed in my work on the Fe-Cr and Ni-B systems, please find them in my publication list. |